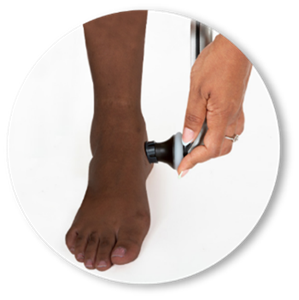
IR-MED to Conduct Usability Study with Methodist Healthcare of San Antonio for its PressureSafe Device Which is Slated for U.S. Launch in H1 2024
- Study to address the substantial challenge of healthcare inequality in the early detection of pressure injuries in people of dark skin tones
- PressureSafe, a skin-color agnostic decision support device, uses AI and infrared light to see beneath skin’s surface to detect biomarkers of pressure injury
-
Interim results from a usability study in Israel indicate PressureSafe identified early-stage pressure injuries with 96% accuracy
Rosh Pina, Israel, Sept. 26, 2023 (GLOBE NEWSWIRE) -- IR-MED Inc., (“IR-MED” or the “Company”) (OTCQB:IRME), developer of a noninvasive artificial intelligence (AI) driven spectrographic analysis technology platform to address significant healthcare needs, announced today it has signed a Clinical Trial Agreement with the Methodist Healthcare System of San Antonio to conduct a useability study titled “Safety and Efficacy of the PressureSafe Device for Early Detection of Pressure Injury in People with Various Skin Tones, Including Dark Skin Tones.” Methodist Healthcare is recognized as the most respected healthcare provider in its region. With a network of 85 hospitals, 9 of which are acute care facilities, Methodist Healthcare employs more than 11,000 people, including 2,700 physicians.
Approximately 50% of subjects recruited for the upcoming study will have dark skin tone, thus producing comparative data on PressureSafe’s accuracy as a decision support device in detecting early-stage pressure injuries in people of darker and lighter skin tones. While early stage pressure injuries can be more difficult to see on dark skin tones, the current standard of care for the detection of pressure injuries is visual skin inspection.
Research shows that people with dark skin tones suffer from pressure injury at more than twice the rate of those with lighter skin tones, as characterized by higher pressure injury rates, higher risks of mortality from pressure injuries, and more severe pressure injuries. According to a study published in September 2023 in Advances in Skin & Wound Care, among all racial and ethnic groups in the U.S., black patients had the highest prevalence of the most severe pressure injuries while white patients had the lowest. The peer-reviewed study goes on to state that due to increasing racial diversity in the U.S., including persons of mixed ethnic and racial backgrounds, there is an urgent need for improved techniques to identify and treat developing pressure injuries earlier.
“We are honored to partner with Methodist Healthcare, a highly prestigious hospital network for our first U.S. usability study of PressureSafe as we prepare for a potential market launch in the first half of 2024, following regulatory filings. The study has been submitted to Methodist’s Institutional Review Board for approval,” commented Dr. Yaniv Cohen, IR-MED’s Co-Founder and Chief Science Officer. “Addressing healthcare equity for the detection of pressure injuries is a priority for IR-MED. By scanning specific bio-markers beneath the skin’s surface using infrared light, we believe PressureSafe can significantly reduce pressure injury related complications regardless of skin tone. Our user-friendly handheld device can improve economics for hospitals, nursing homes, and long-term care providers, while addressing the enormous healthcare burden of this very preventable condition.”
In the U.S. alone, 60,000 patients die every year as a direct result of pressure injuries. Patient care cost per pressure injury ranges from $20,900 up to $151,700, for the 2.5 million patients per year who develop pressure injuries. Pressure injuries are one of the five most common harms experienced by patients and the second most common claim for lawsuits after wrongful death.
IR-MED’s prior usability study for PressureSafe was conducted at two hospitals in Israel owned by Clalit, the world’s second largest health maintenance organization (HMO). Interim results demonstrate PressureSafe detected pressure injuries with 96% sensitivity and 91% specificity. Final results are expected by the end of 2023.
About IR-MED
IR-MED Inc., is developing a noninvasive spectrographic analysis technology platform, allowing healthcare professions to detect, measure and monitor, in real time, different molecules in the blood, in human tissue, and in body fluids without invasive procedures. PressureSafe, the first product under development, is a handheld optical monitoring device that is being developed to support early detection of pressure injuries (PI) to the skin and underlying tissue, regardless of skin tone as it calibrates personally to each patient’s skin.
IR-MED’s technology is being developed to allow accurate readings of biomarkers in a non-invasive method, that may provide caregiver the optimal decision support-system in cases where uncertainties disturb physicians in their decision processes.
IR-MED holds patents protecting its technology and innovations in the noninvasive tissue analysis, and in the modeling and analysis of subcutaneous tissue.
PressureSafe is currently undergoing usability studies at multiple medical centers. It is not yet available for commercial use.
Safe Harbor Statement / Forward-Looking Statements
Statements included in this press release, which are not historical in nature, are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. For example, IR-Med is using forward-looking statements when it discusses the parameters and timing of the study, the expected timing of FDA approval, the potential benefits from its PressureSafe product and the expected timing of the release of its useability study conducted in Israel. Statements relating to the future performance of IR-Med are subject to many factors including, but not limited to, the sufficiency or working capital and our ability to raise the capital needed to fund our development efforts, completion of the development and design of PressureSafe device, results of clinical/useability studies and trials, timing of product development, FDA approval/clearance of products in development, customer acceptance of our products in the market, the introduction of competitive products, the impact of any product liability or other adverse litigation, commercialization and technological difficulties, and the other risks identified in our most recent annual report on Form 10-K filed on March 29, 2023 with the Securities and Exchange Commission. Such statements are based upon the current beliefs and expectations of management and are subject to significant risks and uncertainties. Actual results may differ from those set forth in the forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof, and we do not undertake any obligation to update any forward-looking statements, whether as a result of future events, new information, or otherwise.
Contact:
Sharon Levkoviz, Chief Financial Officer
Tel: +972 (0) 4 6555054
Attachment

Released September 26, 2023